australian curriculum focus
Science / Year 5 / Science Understanding / Chemical sciences / ACSSU077
Content Description: Solids, liquids and gases have different observable properties and behave in different ways
Elaboration:
· recognising that substances exist in different states depending on the temperature
· exploring the way solids, liquids and gases change under different situations such as heating and cooling
Content Description: Solids, liquids and gases have different observable properties and behave in different ways
Elaboration:
· recognising that substances exist in different states depending on the temperature
· exploring the way solids, liquids and gases change under different situations such as heating and cooling
everyday exposure
Links to changes to State of Matter can be made to (examples but are not limited to):
Possibilities to link everyday understanding to solids, liquids & gases are endless. Be creative and link in components relevant to your class.
- Ice melting/freezing water
- Boiling the kettle
- Cooking food
- Eating ice-cream
- Blowing up a balloon etc
Possibilities to link everyday understanding to solids, liquids & gases are endless. Be creative and link in components relevant to your class.
misconception explored
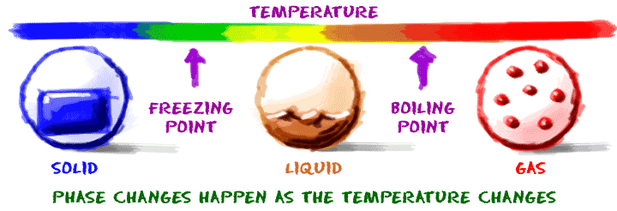
Misconception Three: Boiling is the maximum temperature a substance can reach
Possible reasons why: In general, misconceptions result from students creating their own explanations for how the world works. Often, these ideas are formed well before a student arrives in science class – and serve their purpose well (Fries-Gaither, 2008). Numerous studies and anecdotal evidence show that students cling to these ideas even in the face of discrepant events and explicit instruction. However, some of the misconceptions regarding states and changes of matter can actually be viewed as part of a developmental process. As children age, they are better able to understand states of matter, particularly the more abstract concept of a gas. Students also develop over the years a better understanding of the conservation of matter like water and air. This increasing sophistication of ideas over time should reassure teachers who find that, despite their best instructional efforts, their elementary students still do not fully understand these concepts (Fries-Gaither, 2008).
Misconception explained: Boiling point is the maximum temperature at which the substances is classed as a liquid but, then continues to increase temperature in the form of a gas.
Possible reasons why: In general, misconceptions result from students creating their own explanations for how the world works. Often, these ideas are formed well before a student arrives in science class – and serve their purpose well (Fries-Gaither, 2008). Numerous studies and anecdotal evidence show that students cling to these ideas even in the face of discrepant events and explicit instruction. However, some of the misconceptions regarding states and changes of matter can actually be viewed as part of a developmental process. As children age, they are better able to understand states of matter, particularly the more abstract concept of a gas. Students also develop over the years a better understanding of the conservation of matter like water and air. This increasing sophistication of ideas over time should reassure teachers who find that, despite their best instructional efforts, their elementary students still do not fully understand these concepts (Fries-Gaither, 2008).
Misconception explained: Boiling point is the maximum temperature at which the substances is classed as a liquid but, then continues to increase temperature in the form of a gas.
State of matter
Boiling point: is the temperature at which all of the liquid changes into a gas (or vapour) because the vapour pressure of the liquid is equal to atmospheric pressure. A pure substance has an exact boiling point. Impurities raise (elevate) boiling points (Prescott, 1999). For example adding salt to water.
Freezing point: is the temperature at which all of the liquid changes into a solid. The freezing point of a pure substance is the same temperature as its melting point. Impurities lower (depress) freezing points (Prescott, 1999).
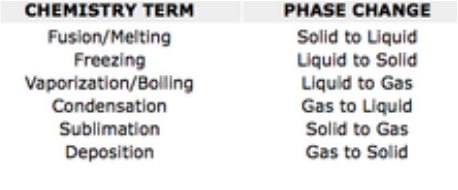
Points of Change
Phase changes happen when certain points are reached. Sometimes a liquid wants to become a solid. Scientists use something called a freezing point or melting point to measure the temperature at which a liquid turns into a solid. There are physical effects that can change the melting point. Pressure is one of those effects (Andrew Rader Studios, 2015) .
When the pressure surrounding a substance increases, the freezing point and other special points also go up. It is easier to keep things solid when they are under greater pressure.
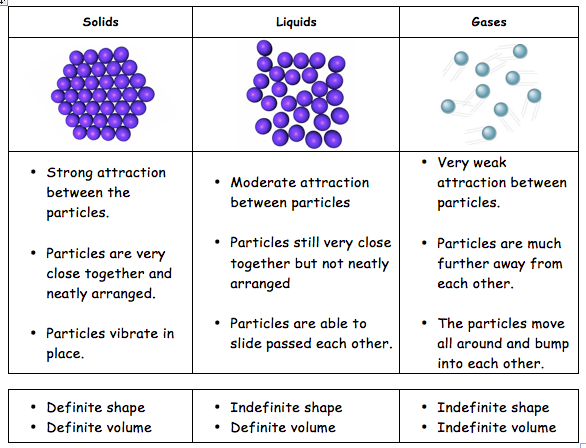
Generally, solids are more dense than liquids because their molecules are closer together. The freezing process compacts the molecules into a smaller space (Andrew Rader Studios, 2015) .
Where to start- unit/lesson plans
|
his comprehensive teacher resource explores the properties of solids, liquids and gases through a series of collaborative inquiry-based learning activities. Students are supported to plan and conduct an investigation of whether the observable properties of materials change with temperature. Seven structured lessons are included, with comprehensive lesson plans, student handouts and additional teaching support material. The unit is based on the 5Es learning model for teaching science, and includes literacy and numeracy focuses to support learning in these areas and assessment strategies relevant to various stages of the teaching unit (Australian Academy of Science, 2012).
|
| whats_the_matter_comp.pdf | |
| File Size: | 5511 kb |
| File Type: | |
Hands On Activities
1. Before viewing
Do you know the temperature that water boils at? What happens to the water at that temperature?
Imagine if water boiled at room temperature.
2. As you view
What is the name of the substance that is added to the large water bottle? Is it a solid, liquid or gas?
Notice the safety equipment worn by the Surfing Scientist and Elliot. Can you name the three items they are using?
3. After viewing
What did you observe happening to the balloon after it was secured to the top of the water bottle?
What was inside the balloon?
The clip demonstrated some of the different properties of liquids and gases. Can you name two?
4. Next steps
Do an internet search to discover the boiling point for liquid nitrogen.
Describe to a friend how the boiling point of nitrogen relates to the balloon pop experiment. Explain why the experiment can be done at room temperature.
Do you know the temperature that water boils at? What happens to the water at that temperature?
Imagine if water boiled at room temperature.
2. As you view
What is the name of the substance that is added to the large water bottle? Is it a solid, liquid or gas?
Notice the safety equipment worn by the Surfing Scientist and Elliot. Can you name the three items they are using?
3. After viewing
What did you observe happening to the balloon after it was secured to the top of the water bottle?
What was inside the balloon?
The clip demonstrated some of the different properties of liquids and gases. Can you name two?
4. Next steps
Do an internet search to discover the boiling point for liquid nitrogen.
Describe to a friend how the boiling point of nitrogen relates to the balloon pop experiment. Explain why the experiment can be done at room temperature.
This video explores the properties of matter and provides a set of exploration questions to accompany the video. The experiment of expanding and shrinking air using a balloon on a bottle can be reproduced as a class experiment with minimal materials needed. Lesson 6 (Hot stuff at a glance) in 'What's the Matter?' in Primary Connections has details for this experiment.



